As the industry’s (more than justified) excitement over last week’s rescheduling announcement begins to subside, the regulatory reality is settling in. In this guest post from regular Business of Cannabis contributor and cannabis policy analyst Deb Tharp, she argues that the Blanche order is not the clean win it appears.
For operators, telehealth platforms and dispensaries rushing to claim the benefits of Schedule III, she has a warning: the same federal jurisdiction that just legitimised your business has also acquired the tools to scrutinise it. Her message to the industry is characteristically direct. The trap has already been set.
You can read the rest of Deb’s insights on the US market, policy, and rescheduling implications here.
The Blanche order formalising Schedule III cannabis is being celebrated across the industry as a major win for medical cannabis – and for truly compliant cannabis operations, it really is a win. 280E tax relief, federal legitimacy, and expedited DEA registration pathways are exciting and undeniable achievements.
However, as always, it’s time for the reality check. This is a federal ‘enough rope’ manoeuvre. It delivers short-term protection and legitimacy for our current state-run recommendation and dispensary systems while quietly building the exact data pipeline the DEA rightfully used to dismantle opioid and ketamine ‘pill mills.’
Cannabis doesn’t deserve similar treatment, but right now, rulemaking and enforcement are in the hands of politicians and bureaucrats who believe it does.
In short, if you run, invest in, or finance a medical cannabis business, you need to keep reading.
For 25+ years, almost all state medical cannabis programs have used a ‘recommendation’ or ‘certification’ system instead of traditional prescriptions. The Blanche order explicitly recognises and temporarily preserves this model.
This is the ‘bait.’ It tells recommendation platforms they don’t need to switch to strict federal prescription pads yet. But it doesn’t exempt them from the Controlled Substances Act (CSA) requirement that every dispensation must have a ‘legitimate medical purpose.’
By validating these certifications federally, the DOJ has also brought them under federal jurisdiction. And this isn’t just a problem for medical practitioners. Medical dispensaries will be feeling the heat too.
This is the mechanism no one is discussing yet under 21 CFR 1306.04(a). This isn’t settled law yet because the rule just came out. However, the reality check here is inevitable. Schedule III status makes ‘corresponding-responsibility’ enforcement a foregone conclusion once certifications are treated as functional prescriptions
How will this look once the system shifts? Dispensaries won’t be allowed to just be a passive vending machine.
The danger isn’t just for those who register with the DEA. This rescheduling scheme creates a pincer movement that captures the entire industry. In a sense, federally registered dispensaries become involuntary informants.
For those who go the DEA route, the data flow is automatic. The feds will be using AI-powered monitoring to track federal registrant records, suspicious order monitoring, inventory records, state seed-to-sale data, and any DEA-imposed reporting pathway. If a dispensary’s purchasing volume spikes or shows a geographic anomaly, it creates a federal ‘heat map.’
Once the DEA audits a high-volume shop, they dive into the state cannabis registry, certification records, PDMP data, where applicable, and dispensary transaction logs. This is where the ‘mill’ is exposed. The DEA will see that a single Telehealth NPI number is responsible for a large percentage of a shop’s certifications, and note other discrepancies — for instance, if a practitioner’s patients are mostly 18-20-year-olds with ‘chronic pain.’
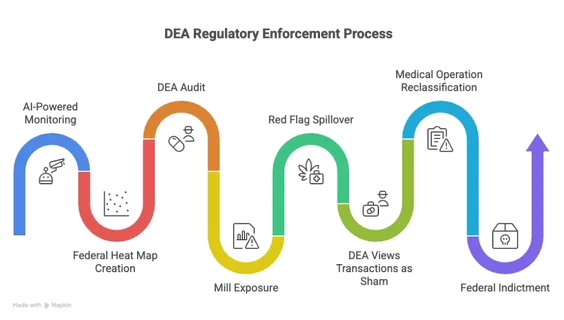
Once the feds become aware of a platform or practitioner, this red flag will spill over even into non-DEA-registered dispensaries that work with them. They aren’t safe, they’re just on a different enforcement path. Under Schedule III, a cannabis sale only has federal protection if it has a ‘legitimate medical purpose.’ If a state-licensed shop pays ‘referral fees’ or offers ‘doctor-specific discounts’ to a platform, the DEA views that transaction as a sham.
In the new regulatory environment, a DEA-registered shop could very well paint a medical operation that was legitimate in 2025 as a ‘mill’ in 2027. If this happens, then a non-DEA-registered shop that is on the mill’s ‘paid referrals’ ledger won’t get a DEA audit. It will get a federal indictment.
Practitioners might argue they are only issuing state ‘certifications,’ not federal prescriptions, so the rules don’t apply to them. The Order attempts to legitimise the system by saying state documents are sufficient, ‘notwithstanding’ normal prescription rules.
Don’t be fooled. That language only validates the format of the paper; it does not excuse the intent of the provider. The underlying federal requirement for a ‘usual course of professional practice’ remains the gold standard.
The DEA has already used this playbook to dismantle the Truepill, Cerebral, and Done ADHD platforms. They didn’t just arrest the doctors; they targeted the pharmacies that ignored the red flags. In the new Schedule III era, the ‘preferred medical platform’ marketing tactics that built the state industry will become the primary evidence for federal conspiracy charges.
Worse, once the data flows from registered cannabis dispensaries, AI pattern-analysis tools will light those red flags up instantly.
Operators are rushing to claim 280E tax relief, which the Order allows even for non-DEA-registered state entities. But be careful.
To claim the deduction, they must file detailed tax returns acknowledging they are a medical cannabis business. If an IRS or DEA audit reveals their ‘medical’ program is actually a ‘recreational mill’ (sham certifications, no follow-up), they lose the Schedule III protection — meaning they’ve just handed the government a signed confession of trafficking Schedule I drug (since they failed the medical test).
And the feds will add tax fraud as an insult to the injury.
The Blanche order is only the first step (a treaty-driven workaround for medical programs). The broader process for full botanical marijuana rescheduling restarts with an expedited administrative law judge hearing on June 29, 2026. That hearing could tighten the recommendation model, add more prescription-like requirements, or change enforcement expectations.
Operators and investors now have a 3–6 month window of relative safety. Use it wisely.
The next 12 months aren’t a victory lap; they are a compliance sprint. The ‘Wild West’ of medical recommendations is over. The federal sheriff is in town, and he’s using your own data to track you.
The post DEA’s Schedule III Cannabis Order: A Free-For-All for Telehealth Platforms, or the Perfect Setup for the Entire Industry? appeared first on Business of Cannabis.
Continue reading...
For operators, telehealth platforms and dispensaries rushing to claim the benefits of Schedule III, she has a warning: the same federal jurisdiction that just legitimised your business has also acquired the tools to scrutinise it. Her message to the industry is characteristically direct. The trap has already been set.
You can read the rest of Deb’s insights on the US market, policy, and rescheduling implications here.
The Blanche order formalising Schedule III cannabis is being celebrated across the industry as a major win for medical cannabis – and for truly compliant cannabis operations, it really is a win. 280E tax relief, federal legitimacy, and expedited DEA registration pathways are exciting and undeniable achievements.
However, as always, it’s time for the reality check. This is a federal ‘enough rope’ manoeuvre. It delivers short-term protection and legitimacy for our current state-run recommendation and dispensary systems while quietly building the exact data pipeline the DEA rightfully used to dismantle opioid and ketamine ‘pill mills.’
Cannabis doesn’t deserve similar treatment, but right now, rulemaking and enforcement are in the hands of politicians and bureaucrats who believe it does.
In short, if you run, invest in, or finance a medical cannabis business, you need to keep reading.
The Recommendation Model Just Got Federally Codified…For Now
For 25+ years, almost all state medical cannabis programs have used a ‘recommendation’ or ‘certification’ system instead of traditional prescriptions. The Blanche order explicitly recognises and temporarily preserves this model.
‘Notwithstanding Part 1306 or any other provision of these rules, a certification or other document (including an electronic document) that state law deems sufficient … shall be sufficient to permit dispensing…‘ (page 29)
This is the ‘bait.’ It tells recommendation platforms they don’t need to switch to strict federal prescription pads yet. But it doesn’t exempt them from the Controlled Substances Act (CSA) requirement that every dispensation must have a ‘legitimate medical purpose.’
By validating these certifications federally, the DOJ has also brought them under federal jurisdiction. And this isn’t just a problem for medical practitioners. Medical dispensaries will be feeling the heat too.
The ‘Corresponding Responsibility’ Trap
This is the mechanism no one is discussing yet under 21 CFR 1306.04(a). This isn’t settled law yet because the rule just came out. However, the reality check here is inevitable. Schedule III status makes ‘corresponding-responsibility’ enforcement a foregone conclusion once certifications are treated as functional prescriptions
How will this look once the system shifts? Dispensaries won’t be allowed to just be a passive vending machine.
The danger isn’t just for those who register with the DEA. This rescheduling scheme creates a pincer movement that captures the entire industry. In a sense, federally registered dispensaries become involuntary informants.
How The Trap Works
For those who go the DEA route, the data flow is automatic. The feds will be using AI-powered monitoring to track federal registrant records, suspicious order monitoring, inventory records, state seed-to-sale data, and any DEA-imposed reporting pathway. If a dispensary’s purchasing volume spikes or shows a geographic anomaly, it creates a federal ‘heat map.’
Once the DEA audits a high-volume shop, they dive into the state cannabis registry, certification records, PDMP data, where applicable, and dispensary transaction logs. This is where the ‘mill’ is exposed. The DEA will see that a single Telehealth NPI number is responsible for a large percentage of a shop’s certifications, and note other discrepancies — for instance, if a practitioner’s patients are mostly 18-20-year-olds with ‘chronic pain.’

Once the feds become aware of a platform or practitioner, this red flag will spill over even into non-DEA-registered dispensaries that work with them. They aren’t safe, they’re just on a different enforcement path. Under Schedule III, a cannabis sale only has federal protection if it has a ‘legitimate medical purpose.’ If a state-licensed shop pays ‘referral fees’ or offers ‘doctor-specific discounts’ to a platform, the DEA views that transaction as a sham.
In the new regulatory environment, a DEA-registered shop could very well paint a medical operation that was legitimate in 2025 as a ‘mill’ in 2027. If this happens, then a non-DEA-registered shop that is on the mill’s ‘paid referrals’ ledger won’t get a DEA audit. It will get a federal indictment.
The ‘Blanche Order’ Illusion
Practitioners might argue they are only issuing state ‘certifications,’ not federal prescriptions, so the rules don’t apply to them. The Order attempts to legitimise the system by saying state documents are sufficient, ‘notwithstanding’ normal prescription rules.
Don’t be fooled. That language only validates the format of the paper; it does not excuse the intent of the provider. The underlying federal requirement for a ‘usual course of professional practice’ remains the gold standard.
The DEA has already used this playbook to dismantle the Truepill, Cerebral, and Done ADHD platforms. They didn’t just arrest the doctors; they targeted the pharmacies that ignored the red flags. In the new Schedule III era, the ‘preferred medical platform’ marketing tactics that built the state industry will become the primary evidence for federal conspiracy charges.
Worse, once the data flows from registered cannabis dispensaries, AI pattern-analysis tools will light those red flags up instantly.
READ MORE…

DEA’s Schedule III Cannabis Order: A Free-For-All for Telehealth Platforms, or the Perfect Setup for the Entire Industry?

Cannabis News Today — Wednesday 29 April 2026: Federal Rescheduling Reshapes Industry as Markets and States Recalibrate

Has Rescheduling Accelerated the Cannabis M&A Wave?
The 280E ‘Paper Trail’ Risk
Operators are rushing to claim 280E tax relief, which the Order allows even for non-DEA-registered state entities. But be careful.
To claim the deduction, they must file detailed tax returns acknowledging they are a medical cannabis business. If an IRS or DEA audit reveals their ‘medical’ program is actually a ‘recreational mill’ (sham certifications, no follow-up), they lose the Schedule III protection — meaning they’ve just handed the government a signed confession of trafficking Schedule I drug (since they failed the medical test).
And the feds will add tax fraud as an insult to the injury.
Waiting for the Other Shoe to Drop: The June 29 Expedited ALJ Hearing.
The Blanche order is only the first step (a treaty-driven workaround for medical programs). The broader process for full botanical marijuana rescheduling restarts with an expedited administrative law judge hearing on June 29, 2026. That hearing could tighten the recommendation model, add more prescription-like requirements, or change enforcement expectations.
Operators and investors now have a 3–6 month window of relative safety. Use it wisely.
What Operators and Investors Should Do Right Now
- Dispensaries should audit platform relationships: Pull patient age breakdowns and explore the legitimacy of the platforms they work with.
- Recommendation platforms should tighten processes: Consider the logistics of a move to video-required encounters and documented medical records everywhere possible. Treat every certification as if a DEA auditor will review it in 2027, because if your patient takes it to a DEA-licensed dispensary, they will.
- Ring-fence operations: Separate medical certification and dispensing lines clearly, especially in dual-license states.
- Prepare records: Smart operators will build systems that go beyond state minimums so they satisfy federal ‘corresponding responsibility’ expectations.
- Get expert help early: Engage pharmaceutical and cannabis-specialised compliance counsel before filing for the expedited DEA registration. Clean applicants will move faster. 6. Watch the June 29 hearing: It will signal what federal enforcement will look like in 2027.
The next 12 months aren’t a victory lap; they are a compliance sprint. The ‘Wild West’ of medical recommendations is over. The federal sheriff is in town, and he’s using your own data to track you.
The post DEA’s Schedule III Cannabis Order: A Free-For-All for Telehealth Platforms, or the Perfect Setup for the Entire Industry? appeared first on Business of Cannabis.
Continue reading...
